
http://www.icr.org/article/carbon-14-found-dinosaur-fossils/

Carbon-14 (or radiocarbon) is a radioactive form of carbon that scientists use to date fossils. But it decays so quickly—with a half-life of only 5,730 years—that none is expected to remain in fossils after only a few hundred thousand years. Yet carbon-14 has been detected in “ancient” fossils—supposedly up to hundreds of millions of years old—ever since the earliest days of radiocarbon dating.”
“Even if every atom in the whole earth were carbon-14, they would decay so quickly that no carbon-14 would be left on earth after only 1 million years. Contrary to expectations, between 1984 and 1998 alone, the scientific literature reported carbon-14 in 70 samples that came from fossils, coal, oil, natural gas, and marble representing the fossil-bearing portion of the geologic record, supposedly spanning more than 500 million years.”“Further, analyses of fossilized wood and coal samples, supposedly spanning 32–350 million years in age, yielded ages between 20,000 and 50,000 years using carbon-14 dating. Diamonds supposedly 1–3 billion years old similarly yielded carbon-14 ages of only 55,000 years.”
“Even (these dates are much) too old when you realize that these ages assume that the earth’s magnetic field has always been constant. (However,) it was stronger in the past, protecting the atmosphere from solar radiation and reducing the radiocarbon production. As a result, past creatures had much less radiocarbon in their bodies, and their deaths occurred much more recently than reported!”
“So the radiocarbon ages of all fossils and coal should be reduced to less than 5,000 years, matching the timing of their burial during the Flood. The age of diamonds should be reduced to the approximate time of biblical creation—about 6,000 years ago.”
https://answersingenesis.org/geology/carbon-14/7-carbon-14-in-fossils-coal-and-diamonds/

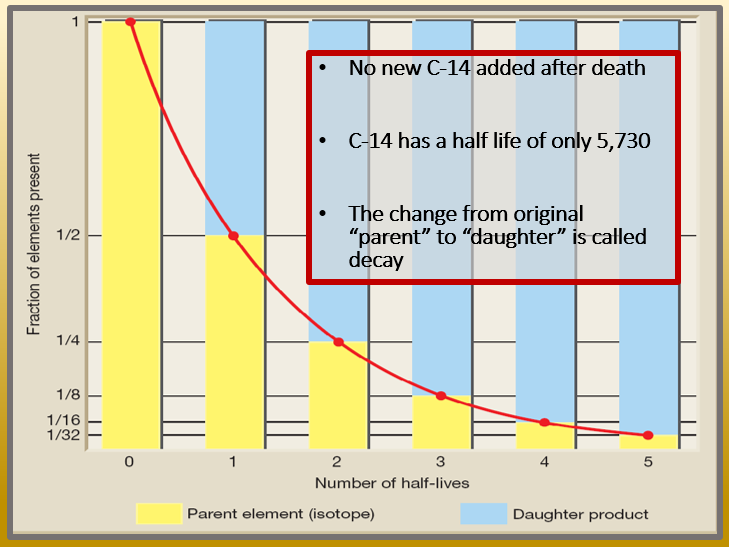
[…] 14, a radioactive isotope of the carbon atom with a known half-life of approximately 5,730 years. A half-life means that half of the original Carbon 14 in any given sample will decay away in about 5,730 years. […]